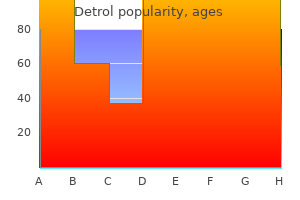
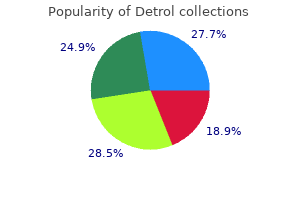
"Order generic detrol on-line, medicine 9312".
W. Murak, M.A., M.D., M.P.H.
Co-Director, University of New England College of Osteopathic Medicine
Wellness Rewards Program Wellness Rewards Program the Plan has a Wellness Rewards Program for members and covered spouses. Each member and their covered spouse can earn a maximum of $250 per calendar year by completing specific activities and screenings. Rewards earned by November 30th of the plan year will be available for use the following calendar year. The reward will be applied to the deductible for members who do not have Medicare B as their primary insurance. Members who have Medicare B as their primary insurance will receive their reward in a health reimbursement account that can be used for qualified medical expenses. For additional information contact the Plan at 866-368-7227, or visit our website at Save 5% to 50% on most dental procedures including routine oral exams, unlimited cleanings and major work such as dentures, root canals and crowns. It delivers an extensive wellness network that focuses on three key components: exercise, eating, and nutrition. New York Life Group Term Life Insurance the Compass Rose Group Term Life Insurance Plan, underwritten by New York Life Insurance Company* helps provide your family with peace of mind. To learn about plan features, costs, eligibility, renewability, limitations and exclusions visit Accident Protection Plan the Compass Rose Accident Protection Plan, underwritten by Nationwide Life Insurance Company, helps provide a financial benefit in case of a death or dismemberment caused by an accident. General Exclusions - Services, Drugs and Supplies We Do Not Cover the exclusions in this section apply to all benefits. There may be other exclusions and limitations listed in Section 5 of this brochure. Although we may list a specific service as a benefit, we will not cover it unless we determine it is medically necessary to prevent, diagnose, or treat your illness, disease, injury, or condition (see specifics regarding transplants). The fact that one of our covered providers has prescribed, recommended, or approved a service or supply does not make it medically necessary or eligible for coverage under this Plan. For information on obtaining prior approval for specific services, such as transplants, (see Section 3 When you need prior Plan approval for certain services). We do not cover the following: Services, drugs, or supplies you receive while you are not enrolled in this Plan. If a provider routinely waives (does not require you to pay) a deductible, copayment or coinsurance, we will calculate the actual provider fee or charge by reducing the fee or charge by the amount waived). Listed below are examples of some of our exclusions: Biofeedback, conjoint therapy, hypnotherapy, milieu therapy, and interpretation/preparation of reports; Charges for completion of reports or forms; Charges for interest on unpaid balances; Charges for missed or canceled appointments; Charges for mailings, faxes, emails or any other communication to or from a hospital or covered provider; Custodial care; Formula unless administered through a tube as the sole source of nutrition; Medical Marijuana; Massage Therapy; 2020 Compass Rose Health Plan 87 Section 6 Mutually exclusive procedures. These are procedures that are not typically provided to you on the same date of service; "Never Events" are errors in patient care that can and should be prevented. If a temporary developmental delay related to a short-term illness or injury occurs, coverage will be subject to normal Plan provisions; Prescriptions for compound powders that have no clinical value; Sales tax for durable medical equipment; Sales tax, shipping and handling for other than durable medical equipment; Select allergy testing, see Section 5(a); Services, drugs or supplies not specifically listed as covered; and Treatment for learning disabilities, developmental delay and mental retardation; The Plan does not cover research costs related to conducting the clinical trial such as research physician and nurse time, analysis of results, and clinical tests performed only for research purposes. Note: Exclusions that are primarily identified with a specific benefit category may also apply to other categories. Note: Refer to Section 5(e) for additional services listed as not covered under mental health and substance abuse benefits. How to claim benefits To obtain claim forms, claims filing advice or answers about our benefits, contact (888) 438-9135, or at our website at Post-service claims procedures We will notify you of our decision within 30 days after we receive your post-service claim. If matters beyond our control require an extension of time, we may take up to an additional 15 days for review and we will notify you before the expiration of the original 30-day period. If you do not agree with our initial decision, you may ask us to review it by following the disputed claims process detailed in Section 8 of this brochure. Records Keep a separate record of the medical expenses of each covered family member as deductibles and maximum allowances apply separately to each person. Save copies of all medical bills, including those you accumulate to satisfy a deductible. If you could not file on time because of Government administrative operations or legal incapacity, you must submit your claim as soon as reasonably possible. If we return a claim or part of a claim for additional information, you must resubmit it within 90 days, or before the timely filing period expires, whichever is later.
Preliminary data suggest that elevated albumin excretion is also a marker of kidney damage in adults with hypertension. Proteinuria in glomerular diseases is primarily due to increased albumin excretion. Therefore, the Work Group concluded that albumin should be measured to detect and monitor kidney damage in adults. The interpretation of albuminuria in kidney transplant recipients is more complicated than in other patients with chronic kidney disease. Second, the main causes of damage in kidney transplant, rejection or toxicity from immunosuppressive drugs, are not characterized by proteinuria. However, diabetic kidney disease is the underlying cause for a large fraction of kidney transplant patients, which may recur in the transplant. Moreover, hypertension is very common after transplantation and is strongly associated with a more rapid loss of kidney function in transplant patients. Finally, recurrent glomerular disease may occur after transplantation and is associated with a greater risk of graft loss. Albuminuria is a better marker than total urine protein of kidney damage due to diabetes, hypertension, and glomerular disease. For these reasons, the Work Group recommends testing and monitoring for albuminuria, rather than total protein, in kidney transplant recipients, as well as in patients with other causes of chronic kidney disease. The cost or technical difficulty of measuring albumin may exceed that for measuring total protein. It is acceptable to measure total protein-to-creatinine ratio as an index of proteinuria in adults when albumin-to-creatinine ratio is substantially elevated (eg, 500 to 1,000 mg/g). However, there is no reliable method to convert ratios of albumin-tocreatinine to total protein-to-creatinine or vice versa. The prevalence of chronic kidney damage due to diabetes and hypertension is far lower in children than in adults. In contrast, the prevalence of kidney disease due to urinary tract abnormalities and congenital tubular disorders is far more common in children than in adults. Evaluation 109 should be measured to detect and monitor kidney damage in most children, one exception being children with diabetes mellitus. The risk of diabetic kidney disease in children is higher in post-pubertal children with duration of diabetes greater than 5 years than in other diabetic children. For these reasons, the American Diabetes Association recommends screening these children for chronic kidney disease, using the same algorithm as for adults. Other diabetic children are screened using the same algorithms as for other children. Excretion of total protein or albumin in the urine are highly variable in individuals with or without kidney disease. Examples of conditions that affect protein excretion other than kidney disease include activity, urinary tract infection, diet, and menstruation. Attempts to avoid these pitfalls include careful definition of events that should preclude the interpretation of abnormal results and consideration of repeat studies when abnormal results are obtained. Some authors have advocated that multiple (up to 5) specimens be obtained in order to obtain a reliable result. However, the Work Group acknowledges the need to repeat abnormal tests, especially low levels of total protein or albumin and the necessity to carefully consider the clinical setting in interpretation of urine protein measurements. A limitation of this guideline is the use of correlation coefficients, rather than more detailed assessments of precision and bias, to assess the accuracy of spot urine measurements of protein-to-creatinine ratios as a measure of protein excretion rates. In addition, other than distinguishing normal from abnormal, the exact level of proteinuria is not usually required for clinical decision-making. Thus, the Work Group concludes that the uniformly high correlation coefficients are sufficiently strong evidence to warrant the conclusions presented here. The relative ease with which proteinuria can be assessed and monitored allows clinicians to identify individuals with completely asymptomatic forms of progressive kidney disease during the early stages of their disease. Such patients may benefit from subsequent changes in management that forestall or prevent additional kidney problems. Proteinuria is a key finding in the differential diagnosis of chronic kidney disease. The relationship between the level of proteinuria and the type (diagnosis) of chronic kidney disease is reviewed in Guideline 6 and in Part 9.
Patients who underwent sex reassignment therapy (both hormonal and surgical intervention) showed improvements in their mean gender dysphoria scores, measured by the Utrecht Gender Dysphoria Scale. This is the largest prospective study to affirm the results from retrospective studies that a combination of hormone therapy and surgery improves gender dysphoria and other areas of psychosocial functioning. A subgroup of the Revision Committee was appointed by the Board of Directors to serve as the Writing Group. From the survey results, the Writing Group was able to discern where these experts stood in terms of areas of agreement and areas in need of more discussion and debate. These decisions were incorporated into the draft, and additional sections were written by the Writing Group with the assistance of the technical writer. The draft that emerged from the consultation meeting was then circulated among the Writing Group and finalized with the help of the technical writer. Feedback from these groups was considered by the Writing Group, who then made further revision. In Western countries, the estimated prevalence of male-tofemale (MtF) transsexualism in community-dwelling adults is about 1 in 10,000 to 1 in 12,000. Among male prison inmates in the United States, the prevalence appears to be significantly higher (Glezer, McNeil, & Binder, 2013). In a study conducted in the California prison system, Sexton, Jenness, and Sumner (2010) interviewed & Anne A. In 2013, the most recent year for which figures are available, there were 135,981 inmates, 95 % of whom were male, in state and federal prisons in California (Carson, 2014); this suggests a prevalence of cross-sex hormone therapy in California inmates of about 1 in 350. The first author, who has served as a consultant to the prison system of a large midwestern state, calculated a prevalence of transgender identification of about 1 in 500 in male inmates, based solely on the transgender inmates she had personally evaluated. These questions and the conflicting opinions they evoke were recently brought into focus by four legal decisions. In December 2014, the entire Court of Appeals for the First Circuit ruled 3 (Kosilek v. This decision was rendered moot in August 2015 when Norsworthy was paroled (``Transgender California inmate,' 2015). If this agreement is carried out, it will represent the first instance we know of in which a U. In the 2001 version, this recommendation was expanded to include other treatments begun before incarceration. Indeed, it is fair to say that this assertion, while admirable in principle, re- mains to be demonstrated in practice in correctional environments. Many have lived troubled, chaotic lives characterized by early family and economic instability, substance abuse and other psychiatric problems, failed school and employment experiences, and early involvement in crime. Is Sex Reassignment Surgery Medically Necessary for Some Inmates With Gender Dysphoria Determining Medical Necessity In the United States, the term``medical necessity'is most commonly encountered in the context of the obligations of third-party payers. For these purposes,``generally accepted standards of medical practice' means standards that are based on credible scientific evidence published in peer-reviewed medical literature generally recognized by the relevant medical community or otherwise consistent with the standards set forth in policy issues involving clinical judgment. Based on expert medical testimony and a review of the published literature, the Appeals Board stated that``We have no difficulty concluding that the new evidence, which includes medical studies published in the more than 32 years since issuance of the 1981 report. We would caution, however, thatthesefavorableconclusions are derived from experience with community-dwelling patients. Although it is reasonable to assume that they would also apply to prison inmates, empirical evidence to support this assumption is lacking. Comorbid psychiatric conditions usually do improve, at least initially, after cross-sex hormone therapy. We believe that many inmates can satisfy all of these requirements withoutunduedifficulty,althoughtheirabilitytofulfilltherequirement of living for 12 months in a gender role congruent with their gender identity remains contentious. For a few inmates, we believe that the 12-month living requirement could legitimately be relaxed or waived. Ofthesix standard eligibilityrequirements, two-ageofmajority and 12 months of continuous cross-sex hormone therapy, the latter with some exceptions permitted-are neither complicated nor controversial. The other standard eligibility requirements involve more complicated considerations as they relate to prison populations. But assessment is not a quick or simple process in either community or correctional settings.
Diseases
- Lecithin cholesterol acyltransferase deficiency
- Progeroid syndrome Petty type
- Aplasia cutis congenita dominant
- Chromosome 9, duplication 9q21
- Mitral valve prolapse
- Cerebellar ataxia infantile with progressive external ophthalmoplegia
- Brachydactyly long thumb type
- Incontinentia pigmenti achromians
- Chromosome 8 ring